A research team led by Professor Fangwei Wang at the Life Sciences Institute, Zhejiang University, has revealed a previously unknown function of the protein Sororin in protecting sister-chromatid cohesion, a fundamental process ensuring accurate chromosome segregation during cell division. Their findings, titled “Sororin locks the DNA-exit gate of cohesin to preserve sister-chromatid cohesion”, are published in Nature Communications, on March 10, 2026.
The study provides a major advance in understanding how cells maintain genomic stability, offering new insights into the molecular machinery that prevents chromosome mis-segregation, a hallmark of cancer and developmental disorders.
A Dual Role for a Key Protein
For years, scientists have known that Sororin helps maintain sister-chromatid cohesion—the physical linkage between duplicated chromosomes—by counteracting a protein called Wapl, which promotes the release of the cohesin ring that holds sister chromatids together. However, whether Sororin plays a more direct role in stabilizing the cohesin ring remained unresolved.
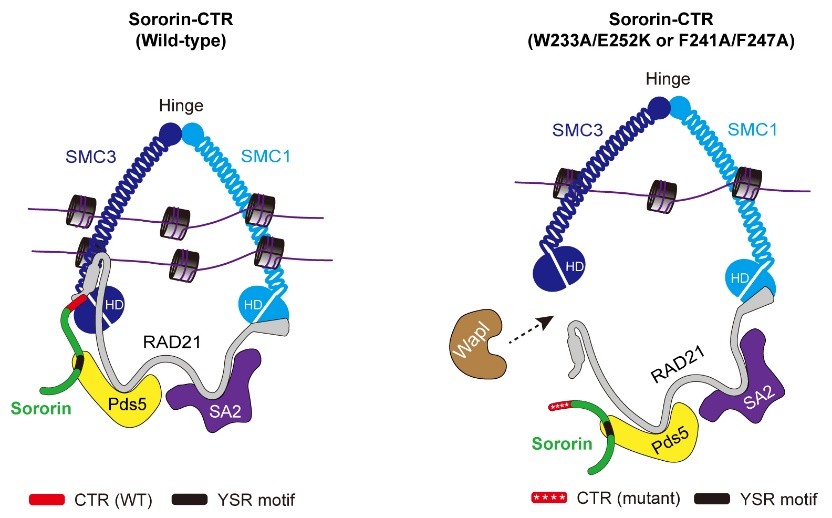
Using a combination of cell biology, biochemical reconstitution, and AlphaFold3-guided structural modeling, the Zhejiang University team demonstrated that the extreme C-terminal region (CTR) of Sororin acts as a “structural lock” that directly binds and stabilizes the DNA-exit gate of cohesin, the interface between RAD21 and SMC3 (Figure 1).
Figure 1. Working model illustrating the function of Sororin’s CTR as a direct structural lock for cohesin’s DNA-exit gate by engaging the RAD21-SMC3 interface. Mutations in Sororin-CTR that disrupt binding to the RAD21-SMC3 interface cause Wapl-dependent opening of the DNA-exit gate and loss of sister-chromatid cohesion.
“We discovered that Sororin does more than just compete with Wapl,” said Professor Wang, the corresponding author of the study. “It physically engages the cohesin ring to lock the gate, ensuring that sister chromatids remain connected until the right moment in mitosis.”
Key Findings
- The Sororin-CTR is both necessary and sufficient to maintain cohesion. When tethered to centromeres, it fully restores cohesion in Sororin-depleted cells.
- Constitutive tethering of Sororin-CTR to chromatin prevents cohesin removal, leading to defective chromosome condensation, impaired decatenation, and segregation errors—phenotypes similar to Wapl loss.
- Aurora B-mediated phosphorylation of Sororin at serine 145 selectively disrupts its interaction with Pds5 (a Wapl-associated factor) while preserving its gate-locking function, providing a “molecular switch” for timely cohesin release in mitosis.
- Specific residues in Sororin, RAD21, and SMC3 mediate this gate-locking interaction, and their mutation abolishes cohesion in a Wapl-dependent manner.
Implications for Disease and Therapy
The study also identifies a cancer-associated mutation (Sororin-E252Q) that weakens cohesin binding, suggesting that defects in gate locking may contribute to genomic instability in tumors.
“Our findings open new avenues for understanding cohesinopathies—developmental disorders caused by cohesin dysfunction—and may inform future therapeutic strategies aimed at modulating cohesin dynamics in cancer,” Wang added.
A Collaborative Effort
The research was led by Drs Qinfu Chen, Xueying Yuan, and Miao Shi as co-first authors, with contributions from Xinyu Zhou, Shukai Zhu, and Drs Weiguo Lu and Haiyan Yan. The work was supported by the Life Sciences Institute at Zhejiang University and reflects the institution’s commitment to advancing fundamental life sciences research with translational potential. The Life Sciences Institute (LSI) at Zhejiang University is a leading research institution dedicated to exploring the fundamental mechanisms of life. LSI fosters interdisciplinary collaboration and cutting-edge research in molecular and cell biology, genomics, and disease mechanisms.
Read the full paper:
https://www.nature.com/articles/s41467-026-70484-2
Chen Q, Yuan X, Shi M. et al. Sororin locks the DNA-exit gate of cohesin to preserve sister-chromatid cohesion. Nature Communications, 2026, 17, 2284. doi:10.1038/s41467-026-70484-2.