On March 13th, 2026, Shixian Lin’s from the Life Sciences Institute, Zhejiang University, published a research article in Nature Chemistry entitled “Recoding multiple rare codons enables the simultaneous incorporation of up to five distinct noncanonical amino acids”.
Expanding the genetic code has revolutionized the ability to study and manipulate biological systems through the site-specific incorporation of noncanonical amino acids (ncAAs). However, current methods in mammalian cells are primarily limited to single-type ncAA incorporation due to translation inefficiency and the exponentially reduced efficiency that occurs when attempting multiplexed incorporation.
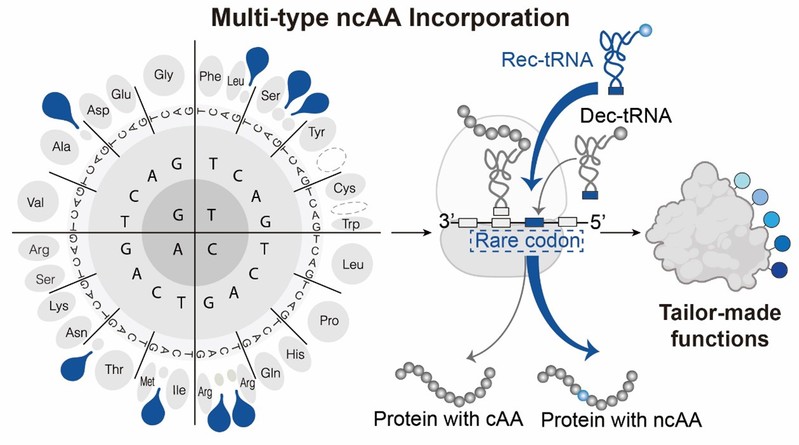
Figure 1. Schematic of the Multi-type Rare Codon Recoding for ncAA incorporation
In this study, the authors developed a multi-type rare codon recoding strategy to address this unresolved challenge. By systematically evaluating the recoding potential of rare codons across the human genome and repurposing them alongside engineered, mutually orthogonal aminoacyl-tRNA synthetase/tRNA pairs, they established a highly efficient multiplexed coding system. They highlight the broad utility of this strategy by achieving the expression of proteins containing two or three distinct ncAAs with recoding rates of up to 90% at wild-type protein expression levels. Furthermore, the system successfully facilitated dual bioorthogonal labelling and sequential protein activation, culminating in the unprecedented synthesis of a single protein incorporated with up to five distinct ncAAs. This breakthrough reveals the redefinable nature of the genetic code and opens entirely new avenues for biomedical and synthetic biology applications.
Yu Fang and Dr. Wei Yu are the co-first authors, Dr. Shixian Lin and Dr. Wenlong Ding are the corresponding authors in this study. This research was supported by the the National Natural Science Foundation of China, the Ministry of Science and Technology of China, the State Key Laboratory of Transvascular Implantation Devices, the Zhejiang Provincial Natural Science Foundation of China, the Feng Foundation of Biomedical Research, and the Fundamental Research Funds for the Central Universities.